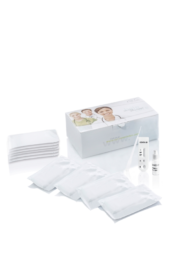
Neutralizing antibody test NADAL® COVID-19 S1-NAb Test från tyska Nal von minden

Ring 042-300 91 30 eller maila info@labteamet.com
Kontrollera om din Covid-19 vaccinering var framgångsrik! testa om du har fått antikroppar efter vaccination.
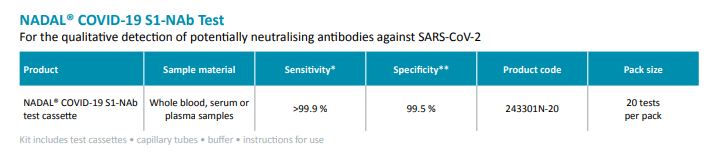
Test för kvalitativ detektion av potentiellt neutraliserande antikroppar mot SARS-CoV-2 i humant helblod, serum eller plasma prover.
Nadal NAB test har hög sensitivitet (>99,9%) och specificitet (99,5%).
Antikroppar kan detekteras, detekterar antikroppar från vaccin som triggar immunresponsen mot S1 enheten av spike-proteinet, såsom de från BioNTech/Pfizer, Moderna®, AstraZeneca, Johnson&Johnson and Sinovac Life Sciences.
Testproceduren är mycket enkel och kräver ingen speciell träning eller kvalifikationer för att utföras. NADAL® COVID-19 S1-NAb Testet är avsett för professionellt bruk.
For the qualitative detection of potentially neutralising antibodies against SARS-CoV-2 in
human whole blood, serum or plasma samples.
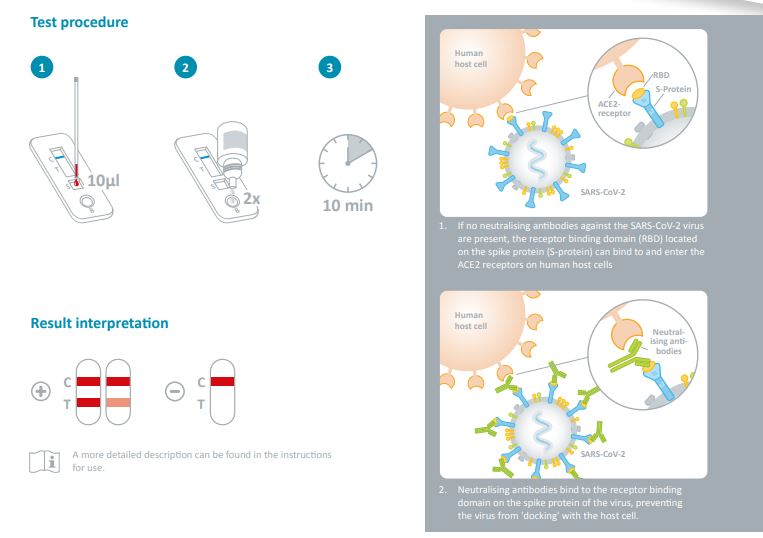
Neutralising antibodies against SARS-CoV-2
The effectiveness of vaccines against coronavirus SARS-CoV-2
lies between 65 % and 95 %*. The detection of neutralising
antibodies can determine whether a vaccine has been successful. Following a vaccination or an infection with coronavirus, various antibodies are produced by the immune system.
However, only some of the antibodies formed have a neutralising effect, i.e. they prevent the virus from entering the host
cell. Neutralising antibodies block the spike protein on the
surface of the virus, preventing it from binding to the host
cell’s ACE2 receptors and thus protecting against a severe
course of infection with COVID-19.
These potentially neutralising antibodies against the S1 subunit of the SARS-CoV-2 spike protein can be detected using a
rapid test.
NADAL® COVID-19 S1-NAb Test
The test serves as an aid in determining whether an immune
response to the S1 subunit of the SARS-CoV-2 spike protein
has occurred. Antibodies can be detected against vaccines
that trigger an immune response against the S1 subunit
of the spike protein, such as those from BioNTech/Pfizer,
Moderna®, AstraZeneca, Johnson&Johnson and Sinovac Life
Sciences. The test procedure is not automated and requires
no special training or qualifications. The NADAL® COVID-19
S1-NAb Test is intended for professional use.
Your benefits at a glance:

| Artikelnummer | 243301N-20 |
|---|---|
| Förpackningsstorlek | 20 testkassetter |
| Varumärke | NADAL® |

LEASING / HYRA / AVBETALNING
Vårt samarbete med Wasa Kredit gör att du snabbt och enkelt får förslag på finansiering som passar dina behov Finansiering upp till 100%. Ni slipper binda ert kapital.
Investera nu i rätt utrustning, för att säkra er konkurrenskraft, kapacitet och tillgång till vital utrustning Sprid kostnaden över den tid som utrustningen används. Utrustningen som säkerhet för finansieringen.







LabTeam Scandinavia AB
Vasatorpsvägen 1G
Godsmottagning – LabTeamet
254 57 Helsingborg
Sweden
LEASING / HYRA / AVBETALNING
Vårt samarbete med Wasa Kredit gör att du snabbt och enkelt får förslag på finansiering som passar dina behov Finansiering upp till 100%. Ni slipper binda ert kapital.
ANMÄL DIG TILL VÅRT NYHETSBREV
Din epost (obligatorisk)
För att ge de bästa upplevelserna använder vi teknik som cookies för att lagra och/eller komma åt enhetsinformation. Genom att godkänna dessa tekniker kan vi behandla data som surfbeteende eller unika ID:n på denna webbplats. Att inte samtycka eller återkalla samtycke kan påverka vissa funktioner och funktioner negativt.
Har du lagt till produkter till ”Mina produkter” så glöm inte skicka iväg din pris- eller produktförfrågan också. Skapa gärna ett konto så sparas alla dina förfrågningarna under ”Mitt konto” sidan.